Have You Been Told to “Wait” After an Abnormal Pap?
You may qualify for a medically supervised women's health study during the “watch and wait” phase in London, Ontario. KGK Science is enrolling women for a 12-month clinical research study evaluating a daily natural health supplement designed to support cervical health.
Up to $1,000 compensation + travel support
7 in-clinic visits + 3 remote check-in
One daily study supplement · simple routine
No medication · No injections
No extra procedures · standard monitoring only
Real Feedback From Patients Who Choose Panacea

Does This Sound Like You?

Many women are told to “wait” after an abnormal Pap result, often with no clear next step.
You may be a fit for this study if you are experiencing:
Abnormal Pap result that hasn’t resolved
HPV that has persisted for 12+ months
Currently in a “watch and wait” phase
Uncertainty about what to do in between appointments
If that sounds familiar, this research study may be worth exploring.
What's Involved
One daily capsule for the study duration (supplement or placebo)
7 in-clinic visits in London, ON + follow-ups
Routine cervical monitoring (Pap & HPV testing)
Questionnaires and health tracking throughout the study
Total duration: approximately 12 months
Up to $1,000 compensation upon completion
(prorated if you exit early · travel costs reimbursed)
You May Qualify If You
Are a female aged 25–55
Have abnormal cervical cells (CIN1+) that haven't resolved
Are HPV positive for 12+ months
Are not currently undergoing treatment
Are not pregnant or planning pregnancy
Can attend clinic visits in London, Ontario
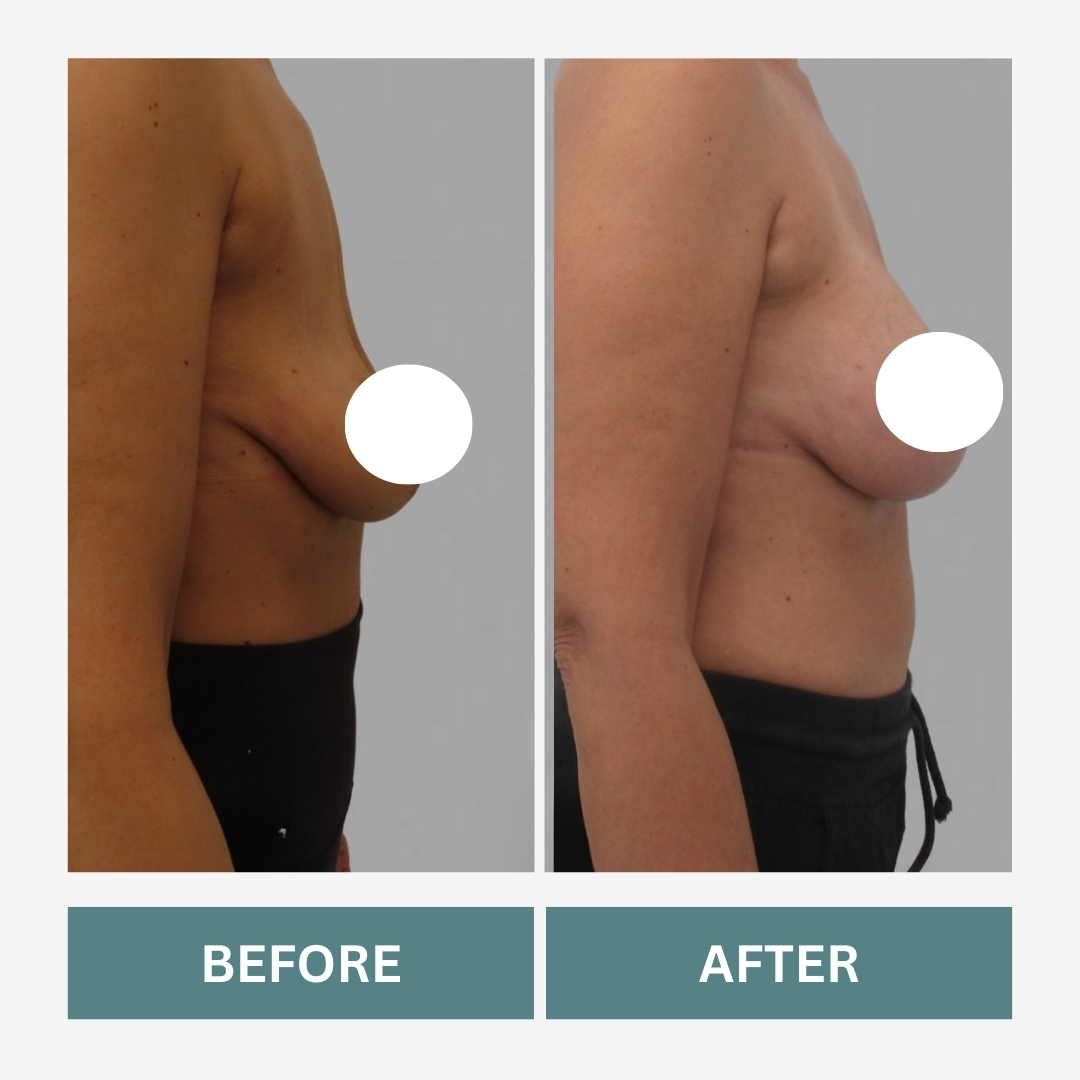
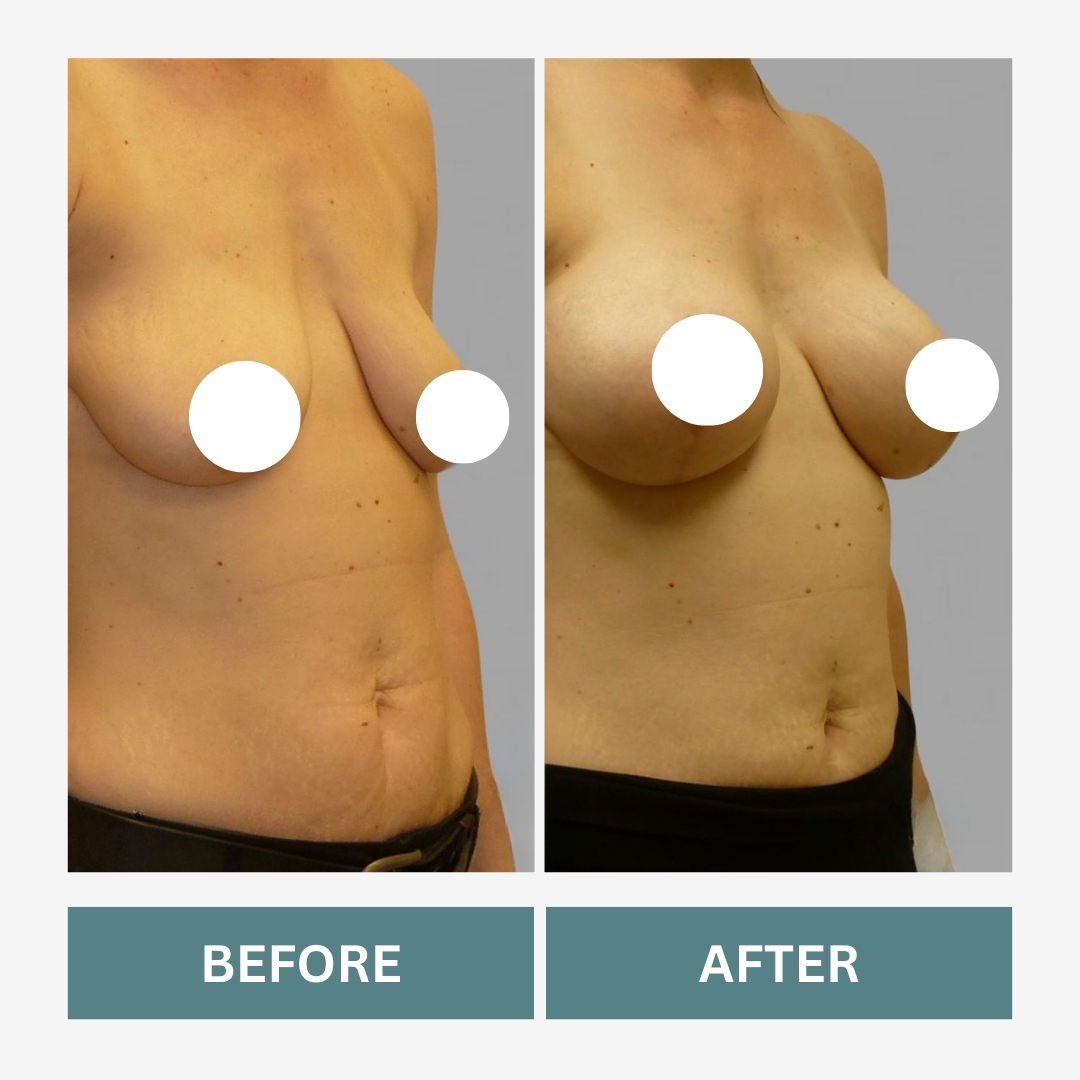
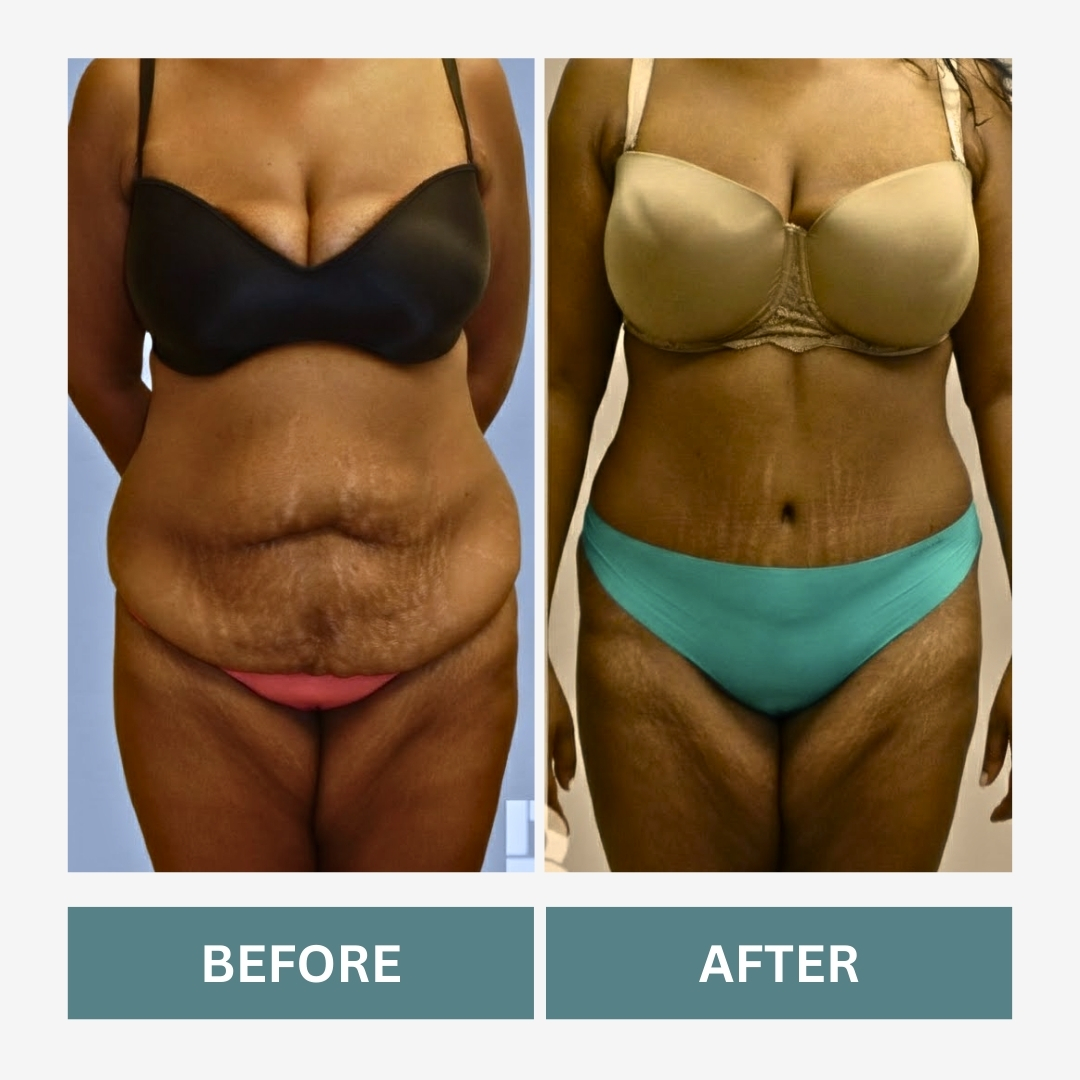
What a Mommy Makeover May Include
A Mommy Makeover is not a single surgery. It combines procedures designed to address post-pregnancy changes.

Tummy Tuck
Removes excess abdominal skin and tightens underlying structure when appropriate.

Breast Augmentation
Restores volume lost after pregnancy or breastfeeding.

Breast Lift
Repositions breast tissue to improve contour.
Consultation, surgery, and recovery are managed within one coordinated care model.

About This Research Study
KGK Science is conducting a clinical research study to evaluate a daily natural health supplement for cervical health.
After an abnormal Pap result and HPV diagnosis, many women enter a monitoring phase where they are told to return for follow-ups while waiting to see how cervical changes progress. During this time, there are often limited options between appointments.
This study aims to better understand how targeted nutritional support may help support cervical health during this period.
The study supplement contains vitamins, minerals, and plant-based compounds studied for their potential role in supporting cellular and immune health, including vitamins A, B12, C, and E, folate, zinc, selenium, green tea extract, broccoli sprout powder, astragalus, lycopene, and reishi mushroom extract.
These ingredients have shown early research interest, but further clinical evaluation is needed.
Why KGK Science
KGK Science has been conducting clinical research for over 27 years.
Study visits take place at:
275 Dundas St, Suite G02 London, ON

Check Your Eligibility in Under 2 Minutes
If you’ve had an abnormal Pap result and are currently in the “watch and wait” phase, you may qualify for this compensated clinical research study.
Frequently Asked Questions
Is this a treatment?
No. This is a clinical research study designed to observe and understand cervical health over time.
Will I be compensated?
Participants may receive up to $1,000 compensation upon completion of the study.
Do I need to stop my current care?
No. Participation is reviewed individually, and the study team will guide you appropriately.
How long is the study?
The study runs for up to 12 months, including visits and follow-ups.
Is my information confidential?
Yes. All personal and medical information is handled with strict confidentiality.
Are there any extra procedures involved?
No additional or experimental procedures are required. All monitoring follows standard clinical practices such as Pap tests, HPV testing, and routine health assessments.
Do I need a formal diagnosis or referral to join?
You do not need a referral, but you must have documentation of an abnormal Pap result or be willing to complete screening tests at the clinic to confirm eligibility.
How do I get started?
Simply complete the short 2-minute eligibility form. If you appear to qualify, the research team will contact you with the next steps.

KGK Science has been delivering top-notch clinical research trials and expert regulatory support for the nutraceutical industry for over 27 years, aiding companies in bringing their products to global markets with custom trials and claim substantiation strategies.
Copyright © 1997-2026 KGK Science Inc.
275 Dundas St., Suite G02
London ON N6B 3L1
Email:- [email protected]
Phone:- (519) 438-9374